In this demonstration, students' assumptions are challenged as they observe the power of surface tension in water droplets.
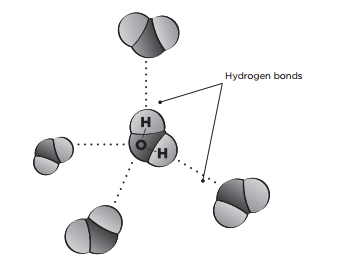
Although a water molecule has an overall neutral charge, the actual structure of a water molecule makes it a polar molecule (it has a positive end and a negative end). The two hydrogen atoms are slightly positive, and the oxygen atom is slightly negative. A weak link occurs between the negatively charged oxygen atom of one water molecule and the positively charged hydrogen atoms of a neighbouring water molecule. This weak link is called a hydrogen bond. The polarity of the water molecule can also cause it to be attracted to molecules of other polar substances.
There are two types of "stickiness" in this demonstration: cohesion and adhesion.
The attraction between water molecules is called cohesion. The cohesive force that occurs between water molecules is so strong that, at the water's surface, it creates a "skin", which is known as surface tension. Surface tension is strong enough to support insects that crawl across the water's surface, like water striders (Gerridae).
The attraction of water molecules to other substances, like soil or glass, is called adhesion. As drops of water are added onto a penny, the adhesive force between the water and the penny keeps the water from falling off.
Cohesive forces are strong, but not unbreakable. As a water drop builds up and out, usually bulging over the sides of the penny, the cohesive forces will eventually be overcome by the force of gravity on the water molecules. The "skin" will burst, and all of the water will spill off.
The cohesive forces between polar molecules are stronger than those between non-polar molecules, such as those in oil or syrup. That's why you can make a bigger "pile" of water than of oil or syrup.