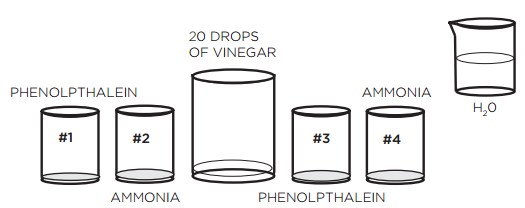
In this activity, students are fooled by an acid-base colour-changing reaction, and learn how chemistry can explain this magical result.
Phenolphthalein is an indicator — a chemical which changes colour depending on whether it meets an acid or a base. It turns purple if it meets something basic, such as ammonia; it stays colourless if it meets an acid like vinegar or a neutral substance like water.
In this magic trick, the phenolphthalein in beakers 1 and 3 will turn purple when you mix it with the ammonia in beakers 2 and 4. When you pour the two purple solutions into the large beaker, the acidic vinegar neutralizes the basic ammonia. The phenolphthalein becomes colourless.
Vocabulary:
- Acid: Acids are compounds with excess H+ ions. The term comes from the Latin word acidus that means “sharp” or “sour”. Some basic properties of acids are that they taste sour, react with metal, change the color of litmus paper from blue to red, and have a low pH (0 – 7). Examples of acids are vinegar and lemon juice.
- Base: A compound that has an excess of OH- ions. Another word for base is alkali. Some basic properties of bases are that they can be used as household cleaners, turn red litmus paper blue, and have a high pH (7 – 14). Some examples of bases are ammonia and baking soda.
- Indicator: A substance that indicates the degree of acidity or alkalinity of a solution through characteristic color changes.