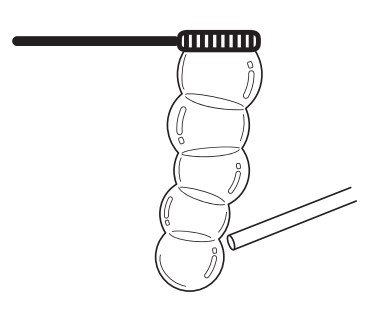
In this activity students use what they know about bubbles to do some impressive tricks. By experimenting with bubbles, students learn about surface tension, elasticity, minimal surface structures, and how to blow really, really big bubbles!
Bubbles fascinate both children and adults with their beautiful shapes and colours. Such simple ingredients—soap and water—create mesmerizing examples of both geometry and chemistry. By experimenting with bubbles, students learn about surface tension, elasticity, minimal surface structures, and how to blow really, really big bubbles!
Soap molecules have two ends, a hydrophobic end and a hydrophilic end. The hydrophobic end avoids water and attaches to oil. The hydrophilic end avoids oil and attaches to water. Soap thus helps attach oily dirt to water so the dirt can be washed away from clothes and dishes.
Surface Tension & Elasticity
Surface tension is the force between liquid molecules at the surface of the liquid (in this case water). The water molecules cling to each other, which causes the surface of the water to behave like an elastic sheet. Elastic substances tend to return to their original shape when they’re stretched.
Water doesn’t make stable, free-floating bubbles all by itself. If you blow a bubble in water, it pops quickly because the surface tension of the water is relatively high and the water is not very stretchy. Adding soap decreases the surface tension so that the water can stay stretched around the bubble.
Even as the bubble is blown up larger or is stretched in various ways, soap stabilizes bubbles by an action known as the Marangoni effect. In places where the bubble is stretched, the surface concentration of soap decreases, which causes the surface tension to increase. This area of increased surface tension springs back to its original form and prevents the bubbles from stretching to the point of popping. The result is spherical bubbles with a uniform surface tension.
Soap also reduces water evaporation so the bubbles last longer, although this effect is relatively small.
Minimal Surface Shapes
When you play with bubbles, you’ll notice that they come in predictable shapes. That’s because deforming a bubble takes energy. The bubble tends to spring back to the shape that is stretched as little as possible – the minimum surface. Floating around in the air, a bubble will become a sphere, because a sphere has the least surface area for a given volume of air. Another way to think about this is that the bubble is stretched most evenly as a sphere (as compared to an egg shape, or a cube). A shape with corners or edges means that the soap is stretched unevenly.